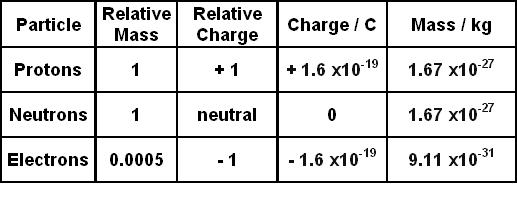
Positive and negative charges can neutralize each other, or neutral particles can split to form positively and negatively charged pairs of particles, but the net amount of charge always remains the same.2.1 Electrons, Protons, Neutrons, and AtomsĪll matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: protons, neutrons, and electrons. That means it cannot be created or destroyed, and the net amount of electric charge in the universe is constant and unchanging. Electrons, though, are believed to be truly fundamental, meaning they cannot be split into smaller parts.Įlectric charge is a conserved quantity. Whenever you try to split a proton or neutron into its constituent quarks, it takes so much energy to do so, the energy is converted into matter in accordance with Einstein’s famous equation E = mc 2, and instead of a single quark, you end up with a neutrally charged quark–antiquark pair, called a meson. However, these particles cannot exist separately. Quantum chromodynamics (QCD) states that protons and neutrons are each composed of three quarks with charges of +2/3 or −1/3 of the unit proton charge, and two of one and one of the other combine to form particles with charges of zero or +1 unit charge. Every accumulation of charge is an even multiple of this number, and fractional charges cannot exist. Protons and electrons carry charges of ☑.602 × 10 −19 C.
Other properties of chargeĮlectric charge is quantized, meaning that it occurs in discrete units. Large accumulations of similarly charged particles are not possible due to their mutual repulsion and their affinity for unlike charges. Just how big of a number is that? Comparing the magnitude of the two forces is like comparing the mass of the Earth to the mass of a single molecule of penicillin! However, gravity still dominates the universe on a large scale, because, unlike charge, it is possible to assemble large quantities of mass. This is true at any distance, since the distance cancels out on both sides of the equation. The repulsive Coulomb force between two protons due to their charge is 4.1 × 10 42 times stronger than the attractive gravitational force between them due to their mass. However, the electrical force is much, much stronger than gravity. The Coulomb force is one of the two fundamental forces that is noticeable on a macroscopic scale, the other being gravity. While this sounds like a small amount, according to HyperPhysics, “Two charges of one coulomb each separated by a meter would repel each other with a force of about a million tons!” Electrical engineers often prefer to use a larger unit for charge, the ampere hour, which is equal to 3,600 C. Coulomb developed the law that says "like charges repel unlike charges attract." A coulomb is defined as the amount of charge transported by a current of one ampere for one second. The unit for measuring electric charge is the coulomb (C), named after Charles-Augustin Coulomb, an 18th-century French physicist.

Therefore, according to the University of Arizona, he invented the terms positive and negative to designate an excess or deficiency, respectively. However, Franklin became convinced that there was only one single electric fluid and that objects could have excess or deficiency of this fluid. Until then, most people thought that electrical effects were the result of mixing of two different electrical fluids, one positive and one negative. Positive and negative values of charge were originally assigned by the American statesman and inventor Benjamin Franklin, who started studying electricity in 1742.
